BACKGROUND
Targeting of the Wnt pathway is an attractive approach to treatment of multiple cancers as it can play a significant role in carcinogenesis. For example, a first-in-class inhibitor (LGK974) of a protein, porcupine, involved in the Wnt pathway has recently completed Phase 1 safety trials. Inhibition of porcupine prevents the palmitoylation of a Wnt signalling protein, an event that otherwise triggers pro-oncogenic outcomes. However, the beneficial impact of this inhibitor in cancer treatment is negated when inactivating mutations to a downstream tumor suppressor protein, FBXW7, are present in a subset of the population. Such alterations in FBXW7 occur at significant frequencies in patients with uterine, colorectal, cervical, and head and neck cancers. Elucidation of the biomolecular consequences of FBXW7 mutations may lead to suggestive mechanisms to treat these subpopulations.
TECHNOLOGY
Researchers at the University of Toronto have found that the presence of FBXW7 mutations or elevated levels of Cyclin1 (CCNL1) in cancer cells can serve as biomarkers to suggest therapeutic intervention using cyclin-dependent kinase 11 (CDK11) or ATR inhibitors. From genome-wide knockouts, they determined that the presence of CCNL1 contributes significantly to the fitness of FBXW7-deficient cancer cells, while its absence does not affect the growth and survival of wild type cells. This suggests that the selective targeting of FBXW7-deficient cancer cells can be achieved through inactivating CCNL1 or perturbing its downstream effects. As CCNL1 is known to interact with CDK11, they further discovered that a known CDK11 inhibitor (i.e. OST964) can re-sensitize FBXW7-deficient cancer cells to therapeutic intervention.
COMPETITIVE ADVANTAGE
- FBXW7 mutations or elevated levels of CCNL1 can serve as biomarkers to guide selective therapeutic intervention
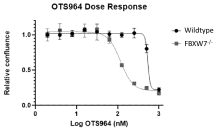
- FBXW7-/- HPAF-II cells show a 5-fold greater IC-50 to a known CDK-11 inhibitor
- FBXW7 mutations occur at frequencies of 18%, 15%, 13% and 8% for uterine, colorectal, cervical, and head and neck cancers respectively
APPLICATIONS
- Biomarker for guiding treatment of FBXW7-deficient cancers
- Relevant cancers include: uterine, colorectal, cervical, and head and neck cancers
INTELLECTUAL PROPERTY STATUS
- PCT application filed (December 2020
PROJECT STATUS
Proof-of-concept studies have been conducted. A genome-wide CRISPR-Cas9 knockout screen was used to identify CCNL1 as a novel substrate of the FBXW7 SCF E3 ubiquitin ligase. This genetic screen was performed in isogenic cell lines (FBXW7 mutant vs wildtype HPAF-II cells). Proliferation and competition experiments were used to validate the synthetic lethal relationship between FBXW7 and CCNL-1. FBXW7-/- cell lines and cervical organoids showed sensitivity to a CDK11 inhibitor (OTS964).
Figure 1. Demonstration of the sensitivity of FBXW7-deficient cells to a known CDK11 checkpoint inhibitor. The chemical structure of the inhibitor is on the left. The dose response curve (right) shows a 5-fold greater sensitivity to FBXW7-/- HPAF-II cells than wildtype, indicating potential for biomarker guidance therapy.