BACKGROUND
Vascular flow delivers nutrients and bioactive compounds and imposes mechanical forces (e.g., shear stress) that shape vessel formation, remodeling, and disease. Yet most human in vitro tissue and organoid systems still rely largely on diffusion or do not recapitulate continuous, physiological intraluminal perfusion at millimeter tissue scales. This limits experimental control over hemodynamics and makes it difficult to model flow-dependent vascular phenotypes in a fully human setting.
TECHNOLOGY
Our researchers have developed VIVOS (for Vascularized In Vitro Organ Systems), an electromechanical, pump-based system for culturing perfusable human vascular beds and enabling perfused delivery into 3D tissues and implanted organoids. The system consists of a magnetically actuated impeller pump connected to a microfluidic device housing a tissue compartment (see Figure 1). A pressure differential between the spanning channel arms (generated by a serpentine resistance channel) drives transverse passage of media through the tissue compartment. In the presence of endothelial and supporting stromal cells in hydrogel, this results in a self-assembled, lumenized vascular network that supports continuous intraluminal flow; with implanted tissues (e.g., cerebral organoids), VIVOS enables perfused delivery and tissue-vessel integration. The pump and microfluidic device use minimal tubing, and three independent units can be accommodated in a standard 6-well SBS format plate, with up to 48 independent units in a standard incubator "half-stack" CO2 incubator.
.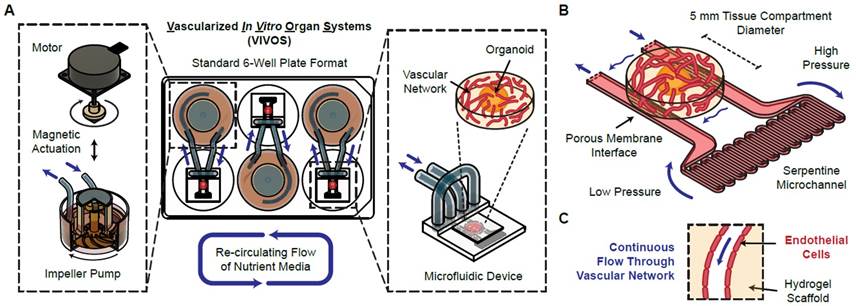
Figure 1. VIVOS platform for electromechanical perfusion of in vitro human vascular beds. (A) The device is composed of a magnetically actuated impeller pump and a microfluidic channel containing a tissue compartment that spans the two arms of the channel. (B) A serpentine microchannel creates a pressure differential in the channel arms, enabling transverse flow through the tissue compartment. (C) This supports formation and continuous perfusion of a vascular network in the absence or presence of implanted 3D tissues (e.g., organoids).
COMPETITIVE ADVANTAGE
- Produces perfusable human vascular beds and supports vascularized organoid/tissue implantation workflows.
- Continuous, tunable, physiologically relevant intraluminal vascular flow (pressure and shear treated as experimental inputs).
- Large ~1 mm × 5 mm × 5 mm tissue compartment compatible with many organoid types and sizes.
- Direct quantitative assays for vascular flow velocities/flow rates, vascular permeability, and vascular morphology in the same bed.
- Optimized modular impeller pump system and microfluidic chip design for reproducibility, robustness, and user friendliness.
- Minimal tubing and high parallelization: 3 units per 6-well plate; up to 48 independent units per incubator.
APPLICATIONS
- Mechanistic vascular biology and mechanotransduction under controllable laminar shear and perfusion.
- Flow-conditioned endothelial remodeling and cell-state transitions (single-cell profiling and perturbations).
- Drug discovery and preclinical evaluation where compound delivery and barrier function can be quantified.
- Human disease modeling in perfused vascular beds (e.g., AVM/HHT-relevant remodeling phenotypes and therapeutic testing).
- Organoid and tissue integration studies requiring perfused delivery (e.g., cerebral organoids and other implanted 3D tissues).
INTELLECTUAL PROPERTY STATUS
- PCT application filed (Sept 2024)
PROJECT STATUS
Prototypes of the device have been built and validated. Endothelial vessels grown in hydrogel display key features including lumenization and deposition of basement membrane components, and support continuous intraluminal perfusion. VIVOS enables quantitative measurement and tuning of vascular hemodynamics and transport, supports implantation of tissues (including human cerebral organoids), and has been applied to single-cell profiling of flow-conditioned vascular remodeling programs and modeling of vascular disease-relevant phenotypes in a fully human setting.





